Human Immunodeficiency Virus (HIV)
BASHH/BHIVA guidelines (see below for individual references).
NICE CKS HIV infection and AIDS. Last revised: Sep 2025.
Background Information
Definition
HIV is a chronic viral infection characterised by progressive immune system damage, primarily targeting CD4+ T lymphocytes, leading to impaired cell-mediated immunity.
Also see the Advanced HIV, AIDS, and HIV-Associated Conditions article.
Virology
HIV is a single-stranded, positive-sense RNA retrovirus [Ref]
- Retroviruses are characterised by the presence of the enzyme reverse transcriptase, which enables conversion of viral RNA into DNA within the host cell
HIV life cycle: [Ref]
| Step | Description | Key involved molecules |
|---|---|---|
| 1. Attachment (binding) | HIV attaches / bind to CD4+ T lymphocytes | Viral gp120 binds CD4 receptor |
| 2. Co-receptor binding | After initial CD4 binding, gp120 changes shape to bind chemokine co-receptors | Viral gp120 binds CCR5 or CXCR4 (chemokine co-receptor) |
| 3. Fusion and entry | Viral envelope fuses with host membrane → viral RNA enters host cytoplasm | Viral gp41 mediates membrane fusion |
| 4. Reverse transcription | Viral reverse transcriptase enzyme converts viral RNA into double-stranded DNA | Viral reverse transcriptase enzyme |
| 5. Integration | Viral DNA integrates into host genome | Viral integrase enzyme |
| 6. Transcription | Host transcribes viral DNA into viral mRNA | Host RNA polymerase |
| 7. Translation | Host translates viral mRNA into viral proteins | Host ribosomes |
| 8. Assembly | Viral RNA and proteins assemble at host cell membrane | Viral polyproteins |
| 9. Budding and release | Immature HIV virion buds from host cell, host membrane is used to form the viral envelope | |
Transmission
HIV can be transmitted from infected body fluids: [Ref1][Ref2][Ref3]
| Body fluids that CAN transmit HIV | Body fluids that CANNOT transmit HIV |
|---|---|
|
|
Important activities that can allow HIV transmission include:
| Sexual activity (vaginal / anal / oral sex) | Transmitted via mucosal membranes
Esp. in the presence of oral diseases (e.g. ulceration, gingivitis) |
| Vertical transmission | From mother to child
During pregnancy, childbirth, or breastfeeding |
| Inoculation | Contaminated needles (e.g. IVDU) / instruments / blood / blood products |
| Direct exposure of mucous membranes or open wounds to infected body fluids | |
| Human bite that breaks the skin |
Importantly, HIV is NOT transmitted by: [Ref]
| Mode of contact | Rationale |
|---|---|
| Social interactions (e.g. hugging, shaking hands) | HIV does NOT penetrate intact skin |
| Sharing eating utensils, glasses, food, and drinks | Saliva contains potent HIV inhibitors and only contains non-infectious viral components |
| Kissing | |
| Toilet seats | HIV does not survive well on environmental surfaces + intact skin barrier |
| Gym equipments | |
| Mosquitoes | HIV does not replicate in mosquitoes |
| Swimming pool and hot tubs | Chlorine inactivates HIV, virus cannot survive in pool water |
Risk Factors
The chance of HIV transmission depends on sexual practice:
- Receptive anal sex > insertive anal sex > receptive vaginal sex > insertive vaginal sex
- Vaginal sex has a lower risk, compared to anal sex
Other risk factors include:
- MSM
- Anal sex practice
- Viral load (the higher the viral load, the more likely the transmission)
- Concurrent STI
- Unprotected sex, sex workers
- IVDU
- Occupational exposure (e.g. sharps and mucosal splash injuries)
- High-risk sexual practice (e.g. chemsex)
- People with a HIV +ve mother
- Blood transfusions, transplants, or other invasive procedures in countries without rigorous HIV screening
Clinical Features
After initial exposure, HIV infection progresses through distinct stages:
| Stage | Clinical manifestation |
|---|---|
| Primary HIV infection / HIV seroconversion illness | Occurs 2-4 weeks after the infection
Patients could be asymptomatic or present with flu-like symptoms:
The viral load is very high in this phase, the patient would be highly infectious. |
| Asymptomatic phase | Follows resolution of the primary infection and may persist for several years
|
| Symptomatic phase | Develops as the patient’s immune function declines
|
| Advanced HIV disease / AIDS | Defined by:
|
Also see the Advanced HIV, AIDS, and HIV-Associated Conditions article.
Reference
Investigation and Diagnosis
Indications for HIV Testing
HIV testing is recommended in:
- People with features consistent with an HIV indicator condition
- People with HIV +ve sexual partners
- People attending the following services
- Sexual health
- Addiction and substance misuse
- Antenatal
- Termination of pregnancy
- Hepatitis B and C, TB and lymphoma
- People commencing chemotherapy or immunosuppressive or immunomodulatory therapy
- People belonging to groups at increased risk of exposure to HIV
- MSM
- Female sexual contacts of MSM
- Black Africans
- Current or prior injecting drug use
- Sex workers
- Prisoners
- Trans women
- From country with high diagnosed seroprevalence (>1%)
- Sexual contact with anyone from a country with high diagnosed seroprevalence (>1%)
- People accessing primary and secondary healthcare in areas of high and extremely high HIV seroprevalence (including emergency departments)
HIV Diagnostic Pathway
Initial screening test: 4th-generation combination assay (HIV p24 antigen + HIV antibody) with venous sampling ASAP after potential exposure
Action / interpretations:
| +ve Screening test | Immediately perform a confirmatory assay on a different sample (ideally, a fresh blood sample)
|
| -ve Screening test | Subsequent action depends on whether the testing is performed within the 45-day window period or not
See greenbox for rationale. |
If results are +ve from self-tests, confirmatory laboratory testing is always required due to a small possibility of a false-positive result.
Rationale:
The 4th-generation combination assay has a window period of 45 days.
The window period is the time between HIV exposure and when the test can reliably detect infection:
- <45 days post-exposure → test may be falsely negative → repeat testing required at ≥45 days
- ≥45 days post-exposure → a negative test can reliably exclude HIV
Baseline Post-Diagnosis Investigations
The following tests are recommended at baseline (organised into the following 3 categories):
| Category | Tests |
|---|---|
| HIV-related tests |
|
| Infection screen |
|
| Routine bloods |
|
References
HIV Management – Antiretroviral Therapy (ART)
Indications and Timing
Choice of Drugs and Regimen
HIV is treated with antiretroviral therapy (ART), a treatment regimen typically comprised of 3 or more different antiretroviral drugs to overcome the risk of drug resistance.
HIV-1 (most cases in the UK)
There are two 1st line regimens:
| Triple therapy (most commonly used) | 2 NRTIs PLUS 1 integrase inhibitor
|
| Two-drug ART regimen |
|
Pre-testing for HLA-B*57:01 is necessary before starting abacavir.
Patients who are HLA-B*57:01 positive must NOT receive abacavir, as they are at risk of a severe hypersensitivity reaction.
Tenofovir DX (disoproxil) is an older oral pro-drug, while tenofovir AF (alafenamide) is a newer oral pro-drug.
Tenofovir DX carries a higher risk of renal and bone toxicity than tenofovir AF.
HIV-2 (rare in the UK, endemic in West Africa)
HIV-2 has intrinsic resistance to all NNRTIs, therefore it is important to avoid them.
1st line: triple ART with 2 NRTI backbone and boosted protease inhibitor
- Tenofovir / abacavir (NRTI) + lamivudine (NRTI) + boosted darunavir (protease inhibitor)
Ongoing Follow-up and Monitoring
- Minimum yearly review
- Aim for viral load of <50 copies/mL
References
HIV PrEP (pre-exposure prophylaxis)
HIV Testing in PrEP
3-monthly HIV testing should be routinely offered as part of monitoring for PrEP.
Indications
Main indications are:
-
Condomless receptive anal sex (MSM and trans-women)
-
Condomless sex with HIV +ve partner with VL >200 copies/mL
-
IVDU with shared equipment (not routinely recommended if effective needle-exchange or opioid substitution programmes exist)
Drugs and Regimen
1st line regimen (dual NRTI): tenofovir disoproxil + emtricitabine (300/200 fixed dose combo)
There are 2 main dosing regimens:
| Dosing regimen | Suitability | Description |
|---|---|---|
| Daily PrEP | All high-risk individuals | One tablet daily → until 7 days after last exposure |
| On-demand PrEP | Anal exposures ONLY | 2+1+1 (2 tablets pre-sex → 1 tablet 24 hr post sex → 1 tablet 48 hr post sex)
If multiple-day exposure → 1 tablet daily until 48 hr after last exposure |
It is important to ensure the person is HIV -ve before taking PrEP. As PrEP regimens contain only 2 drugs, if the patient is already HIV +ve, using only 2 agents risks sub-therapeutic suppression (allowing the virus to replicate) and drug resistance.
Reference
HIV PEP (post-exposure prophylaxis)
Indications
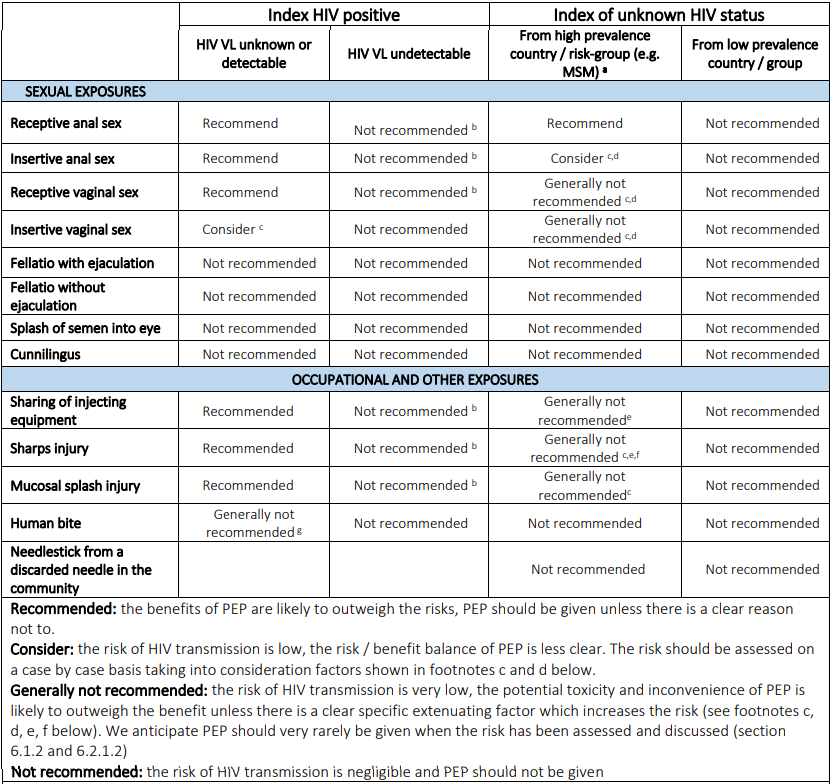
Indications for PEP are categorised and presented below:
| Type of exposure | Indications |
|---|---|
| Sexual exposure | If index HIV +ve
If the index HIV status is unknown
|
| Occupational exposure | Only indicated if index HIV +ve AND any of the following:
|
Key points about the risk of transmission:
- Receptive sex carries a higher risk than insertive sex.
- Anal sex carries a higher risk than vaginal sex.
BASHH/BHIVA – Summary table of PEP prescribing recommendations
Baseline Tests
Baseline tests required before initiating PEP:
- HIV-1 Ag/Ab
- Serum creatinine and eGFR
- Alanine transaminase
- Hepatitis B serology – if not vaccinated or with documented HepBsAb >10 IU
- Chlamydia, gonorrhoea, syphilis testing – if from sexual exposure
- Hepatitis C screening – if from occupational exposure OR those at risk from sexual exposure (e.g. MSM)
Drugs and Regimen
1st line regimen: 3 drug tenofovir (NRTI) + emtricitabine (NRTI) + raltegravir (integrase inhibitor)
Timings:
- Start PEP within 72 hours (ideally <24 hours, guidelines say not to initiate PEP if >72 hours)
- Duration of PEP: 28 days (4 weeks)
Follow Up
Repeat HIV testing at 45 days after completion of the 28‑day PEP course (not 45 days after exposure).
Reference
HIV in Pregnancy
See the HIV and Pregnancy article.