Osteoporosis
NICE clinical guideline [CG146] Osteoporosis: assessing the risk of fragility fracture. Last updated: Feb 2017.
NOGG Clinical guideline for the prevention and treatment of osteoporosis. Last updated: Dec 2024.
NICE BNF Treatment summaries. Osteoporosis.
Background Information
Definition
Osteoporosis is defined by low bone mass and microarchitectural deterioration of bone tissue
Objectively, osteoporosis can be defined by measuring bone mineral density (BMD) using dual energy X-ray absorptiometry (DEXA):
- T score less than -2.5: osteoporosis
- T score between -1.5 and -2.5: osteopenia
T-score vs Z-score
DEXA scans report both a T-score and a Z-score.
Definitions:
- T-score: number of standard deviations by which a patient’s BMD differs from the mean BMD of a healthy young adult reference population, typically aged 20–30 years.
- Z-score: number of SDs by which a patient’s BMD differs from the mean BMD of an age-, sex-, and ethnicity-matched reference population.
Clinical use
- T-score → Main score used to definitively diagnose osteoporosis in adults – especially useful in post-menopausal women and men ≥50.
- Z-score → Cannot definitively diagnose osteoporosis in adults, but is indicated for premenopausal women, men <50, and children, to identify whether BMD is lower than expected for age and to prompt evaluation for secondary causes of osteoporosis if the Z-score is ≤-2. [Ref]
Risk Factors
Lifestyle-related risk factors:
- Post-menopausal women
- Advancing age
- Inadequate physical activity
- Low body weight
- Smoking and excess alcohol consumption
Secondary causes:
| Medications |
|
| Medical conditions |
|
Clinical Features
Osteoporosis on its own is asymptomatic.
Osteoporosis is only clinically noticeable, when a fragility fracture occurs.
- Fragility fractures are fractures that occur from a low-energy mechanism
- Fragility fractures should be suspected in adults (especially postmenopausal women and older men) who present with fractures after minimal trauma, such as a fall from standing height, or who have radiographic evidence of vertebral compression fractures, unexplained height loss, or kyphosis
- They should also be considered in younger patients with risk factors for secondary osteoporosis, such as chronic glucocorticoid use, hypogonadism, or metabolic bone disorders
Assessment and Decision Algorithm
Disclaimer:
The structure of this section has been intentionally designed to support learning, revision, and exam-style application. At first glance, the layout may appear different from the way NICE and NOGG present their recommendations, as the information has been reorganised to improve clarity and usability.
For exam purposes, it is important to recognise the following few points:
- BMD <-2.5 (i.e. osteoporosis) → bone-sparing treatment is indicated
- One may notice that this is NOT included in the guidelines / section below, as BMD <-2.5 alone is NOT an indication to treat
- Once BMD is available, it should be used to recalculate the 10-year risk of fragility fracture and plot it against the NOGG intervention threshold to guide management
- However, in exams one would be expected to know that an osteoporotic BMD (<-2.5) would necessitate bone-sparing treatment (as it will almost certainly put you above the treatment threshold)
- Following a fragility fracture (in post-menopausal women, and men >50 y/o) → start bone-sparing treatment immediately
- Patients starting high-dose oral glucocorticoids (≥7.5 mg/day prednisolone or equivalent over 3 months) → start bone-sparing treatment at the same time as glucocorticoid therapy (without awaiting DEXA scan or calculating FRAX)
If all of the above do NOT apply → calculate the 10-year fragility fracture risk (with FRAX / QFracture) to inform management (i.e. the standard pathway)
Step 1 – Exclude Alternative Causes
Always exclude the following first:
- Secondary causes of osteoporosis (via routine bloods)
- Non-osteoporotic causes of fracture (e.g. Paget’s disease, bone metastases, multiple myeloma)
Step 2 – Assess Need For Bone-Sparing Treatment
Standard Pathway (Most Patients)
This applies to ANY of the following:
- ALL women >65 y/o and men >75 y/o
- Women <65 y/o and men <75 y/o with risk factors
- <50 y/o only with major risk factors (previous fragility fracture / untreated premature menopause / current or frequent use of oral steroids)
The pathway is as follows:
- Calculate the 10-year risk of fragility fracture
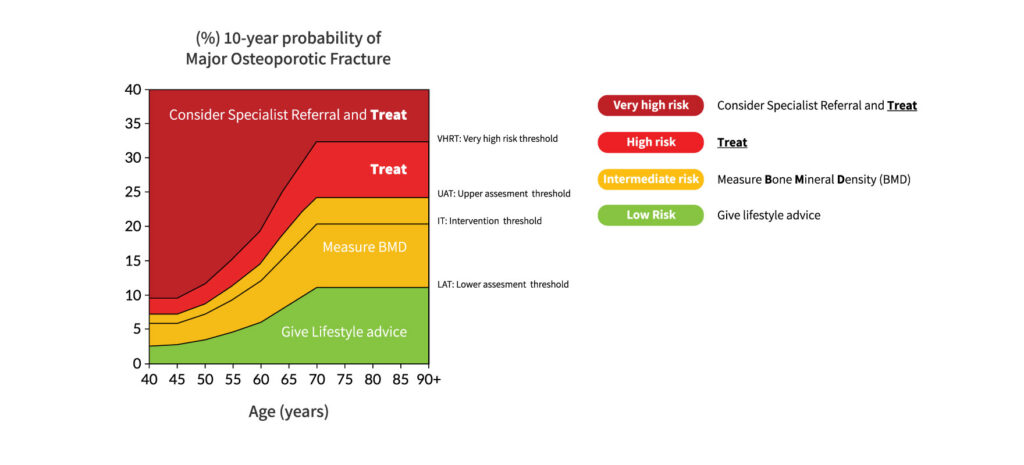
- Plot the 10-year risk (%) against age into the NOGG intervention and risk thresholds (see image below)
- Subsequent actions depend on the risk category
| Risk | Subsequent action |
|---|---|
| Low risk |
|
| Intermediate risk |
|
| High risk |
|
| Very high risk |
|
Exceptions (The High-Risk Population)
There are 3 main exceptions:
| Following a fragility fracture (in post-menopausal women, and men >50 y/o) |
|
| Starting high-dose oral glucocorticoids (≥7.5 mg/day prednisolone or equivalent over 3 months) |
|
| Starting treatment that may have a rapid adverse effect on bone density
Notable treatments include androgen deprivation therapy for prostate cancer and aromatase inhibitors for breast cancer |
|
Management
Conservative / General Management
The following should be given to ALL patients (even those with low fracture risk):
| Lifestyle advice |
|
| Calcium intake |
|
| Vitamin D intake |
|
Bone Sparing Treatment
Choice of Bone-Sparing Treatment
BNF-oriented recommendations:
| 1st line | Oral bisphosphonates
|
| 2nd line (usually if oral bisphosphonates are contraindicated or not tolerated) |
|
| 3rd line (specialist only) |
|
It is worth noting that NOGG recommends annual IV zoledronic acid infusion as 1st line therapy in patients with osteoporosis following a hip fracture
Anabolic agents (teriparatide or romosozumab) may be considered for very high-risk patients; they are typically initiated by specialists
- Definition of very high-risk patients: recent vertebral fracture (within the last 2 years) or ≥2 vertebral fractures or BMD T-Score ≤−3.5
- Anabolic therapy should be followed by anti-resorptive treatment (bisphosphonate or denosumab) to maintain the gains in bone mineral density
Overview of Bone-Sparing Treatment
| Category | Drug | MoA | Route of Administration | Schedule |
|---|---|---|---|---|
| Anti-Resorptive | Bisphosphonates (Alendronic acid, Risedronate, Zoledronic acid) | Inhibit osteoclast-mediated bone resorption | Oral (alendronic acid, risedronate)
IV infusion (zoledronic acid) |
Alendronic acid, risedronate: once weekly OR once daily
Zoledronic acid: 5 mg infusion once yearly |
| Denosumab | Monoclonal antibody against RANKL → inhibits osteoclast | Subcutaneous injection | 6 monthly | |
| Raloxifene | Selective estrogen receptor modulator (agonist in bone → reduces resorption) | Oral | Once daily | |
| Strontium Ranelate (limited use due to safety concerns) | Dual action: stimulates osteoblast bone formation and reduces osteoclast activity | Oral | Once daily | |
| Anabolic | Teriparatide | Recombinant parathyroid hormone → activates osteoblast | Subcutaneous injection | Daily for up to 24 months |
| Romosozumab | Monoclonal antibody against sclerostin → increases bone formation and reduces resorption | Subcutaneous injection | Once monthly for 12 months |
Information on Specific Bone-Sparing Treatments
Information on bisphosphonates is by far the most important and high-yield one to learn. Denosumab is also included as it’s safety profile is similar to bisphosphonates, and is a commonly used 2nd line bone-sparing treatment.
Bisphosphonates Therapy
Administration Instructions
The following applies only to oral bisphosphonates:
- The tablet should be taken after an overnight fast
- Take the tablet at least 30 min before the first food or drink or any other medications / supplementation (including calcium and vitamin D)
- Swallow the tablet with a glass of plain water, while the patient is sitting or standing in an upright position
- Do NOT lie down (stay in sitting or standing position) for 30-60 min after taking the tablet
- Alendronate and risedronate: 30 min
- Ibandronate: 60 min
Adverse Effects and Complications
| Common | With oral tablets:
IV drug form mainly causes an acute phase reaction 24-72 hours after the infusion:
|
| Rare but serious (both oral and IV drug forms) |
|
Caution and Contraindications
There are 2 main things to consider before starting bisphosphonates:
| Hypocalcaemia |
Rationale: Bisphosphonates inhibit osteoclast-mediated bone resorption, which ↓ release of calcium from bone into the circulation. If bisphosphonates are started in someone with low baseline calcium (or vitamin D), this can precipitate hypocalcaemia. |
| Renal impairment | Assess renal function before starting bisphosphonates:
|
| Oesophagus abnormalities | Examples include:
|
| Inability to stand or sit upright for at least 30-60 min | Examples include:
|
Follow Up
Initial Follow Up
- Check treatment tolerance after 3-4 months + ask about adverse effects
- Check adherence after 12 months of treatment
Long-Term Following Up (Continuing or Pausing Treatment)
Reassess the need for continuing treatment after:
- 5 years of oral bisphosphonate therapy, or
- 3 years of IV bisphosphonate therapy)
Reassessment procedure:
- Perform FRAX (or QFracture) with BMD included
- Plot the 10-year risk (%) against age into the NOGG intervention and risk thresholds
- Subsequent action
- Lower risk (i.e. below the intervention threshold) → consider treatment pause for 1.5-3 years (‘drug holiday’) to reduce the risk of atypical fractures and osteonecrosis of the jaw (which are associated with long-term bisphosphonate therapy)
- Above the intervention threshold → continue drug treatment
Exception: high-risk patients require treatment continuation for ≥10 years (oral bisphosphonates) or ≥6 years (IV bisphosphonates), irrespective of reassessment.
High-risk patients are defined as ANY of the following:
- ≥70 y/o at the start of bisphosphonate treatment
- A previous hip or vertebral fracture
- Experienced a further fragility fracture during the initial 5 years of treatment
- On high-dose oral glucocorticoids (≥7.5 mg prednisolone/day or equivalent)
- In these patients, treatment is typically continued until oral steroids are stopped (risk reassessment done thereafter)
Denosumab Therapy
Adverse Effects and Complications
Similar to bisphosphonates, denosumab is also associated with the following rare but serious complications:
- Osteonecrosis of the jaw
- Atypical femoral fracture
Long-Term Follow-Up and Stopping Denosumab
Key considerations:
- All patients should have calcium checked prior to each dose
- Denosumab can cause / worsen hypocalcaemia
- Avoid unplanned cessation of denosumab
- After denosumab is stopped, it causes a rebound in bone mineral density reduction → increased risk of multiple vertebral fractures
- If denosumab therapy is stopped, an IV infusion of zoledronate is recommended 6 months after the last injection of denosumab
References